Subject-Matter Eligibility at the EPO: Life Sciences
January 25, 2021
Authored and Edited by K. Victoria Barker, Ph.D.; Maeve O'Flynn
Subject-matter eligibility at the USPTO for life sciences inventions can be a minefield, and in recent years, numerous articles and webinars have tried to get a handle on what sort of claims might be allowed by the USPTO (our most recent webinar on this topic may be found here). Happily, subject-matter eligibility is much less controversial at the EPO, and the case law is generally consistent, although not entirely without U-turns. In this blog post, we summarise the types of claims that are acceptable at the EPO and look at ways of minimising future subject-matter eligibility issues when drafting.
Exclusions
The EPO does not allow inventions that are contrary to “ordre public” or morality when commercially exploited.[1]More specifically, this excludes patents relating to:
(a) processes for cloning human beings;
(b) processes for modifying the germ line genetic identity of human beings;
(c) uses of human embryos for industrial or commercial purposes; and
(d) processes for modifying the genetic identity of animals which are likely to cause them suffering without any substantial medical benefit to man or animal, and also animals resulting from such processes.[2]
The main caveat to be aware of here is that, while uses of human embryos are not subject-matter eligible, EPO case law allows claims relating to the use of human embryos if (i) the application has a priority or filing date on or after 5 June 2003, and (ii) its technical teaching can be put into practice using human embryonic stem cells derived from parthenogenetically activated human oocytes.
The European Patent Convention (EPC) also prohibits patenting of plant and animal varieties or essentially biological processes for the production of plants or animals, although this prohibition does not extend to microbiological processes.[3] The meaning of the term “essentially biological processes” has been the subject of much debate at the EPO in recent years. However, in May 2020, the Enlarged Board of Appeal clarified that this prohibits product claims and product-by-process claims directed to plants, plant material or animals, if the claimed product is exclusively obtained by means of an essentially biological process or if the process features define an essentially biological process.[4]
Of note here is that the EPO Boards of Appeal have historically been more lenient when considering whether a process for the production of animals is “essentially biological”. Consequently, animals obtained by genetic manipulation seem likely to remain patentable, but animals obtained via selective breeding methods will not. The most obvious reason for this is that, for plant varieties, registration with the Community Plant Variety Office provides an alternative to patent protection in Europe, but no such alternatives exist for animal varieties.
The final exclusion to consider in the life sciences field is “discoveries”.[5]However, in practice, this is a narrow exclusion as it only prohibits “mere discoveries” and not discoveries that have a technical effect. The EPO Guidelines for Examination 2019 gives the example of a substance occurring in nature which is found to have an antibiotic effect. Since an antibiotic effect is a technical effect, such a substance may be patentable.[6]
For the specific field of gene technology, it is a requirement that the industrial application of a sequence or a partial sequence of a gene be disclosed in the patent application.[7]“Industrial application” in this context simply means the purpose or effect of the gene sequence and is intended to prohibit wholly speculative patent applications.
Medical Methods
It is well known that the EPO does not allow “method of treatment” style claims. It is worth noting, however, that the wording of the EPC actually prevents the patenting of methods of medical treatment on the human or animal body by surgery or therapy, and diagnostic methods practised on the human or animal body.[8]
As used by the EPO, the term “therapy” means the treatment or prevention of a disease state. Consequently, cosmetic methods are not therapeutic and are not excluded from patentability. Interestingly, the EPO does not consider that contraceptive methods are therapeutic either since, as noted in the case law, “pregnancy is not an illness”. In veterinary medicine, non-therapeutic and non-cosmetic treatments may be administered to livestock to improve a desirable characteristic, such as milk yield. In such cases, method claims have been allowed, provided that they are clearly marked as “non-therapeutic”.
While “method of treatment” style claims can usually be redrafted as “medical use” claims, amending surgical and diagnostic method claims for conformity with EPO practice can be more difficult. In particular, removing features from originally filed claims at the EPO is increasingly difficult given the very strict added matter requirements. We therefore recommend that, when drafting applications relating to surgical or diagnostic methods, claims without active steps carried out on the body are also included. For example, the first step in the diagnostic method may be “providing a sample” rather than “obtaining”. It may also be worthwhile including EPO-style medical use claims, e.g., a substance or composition for use in a diagnostic method/ surgical method.
Summary
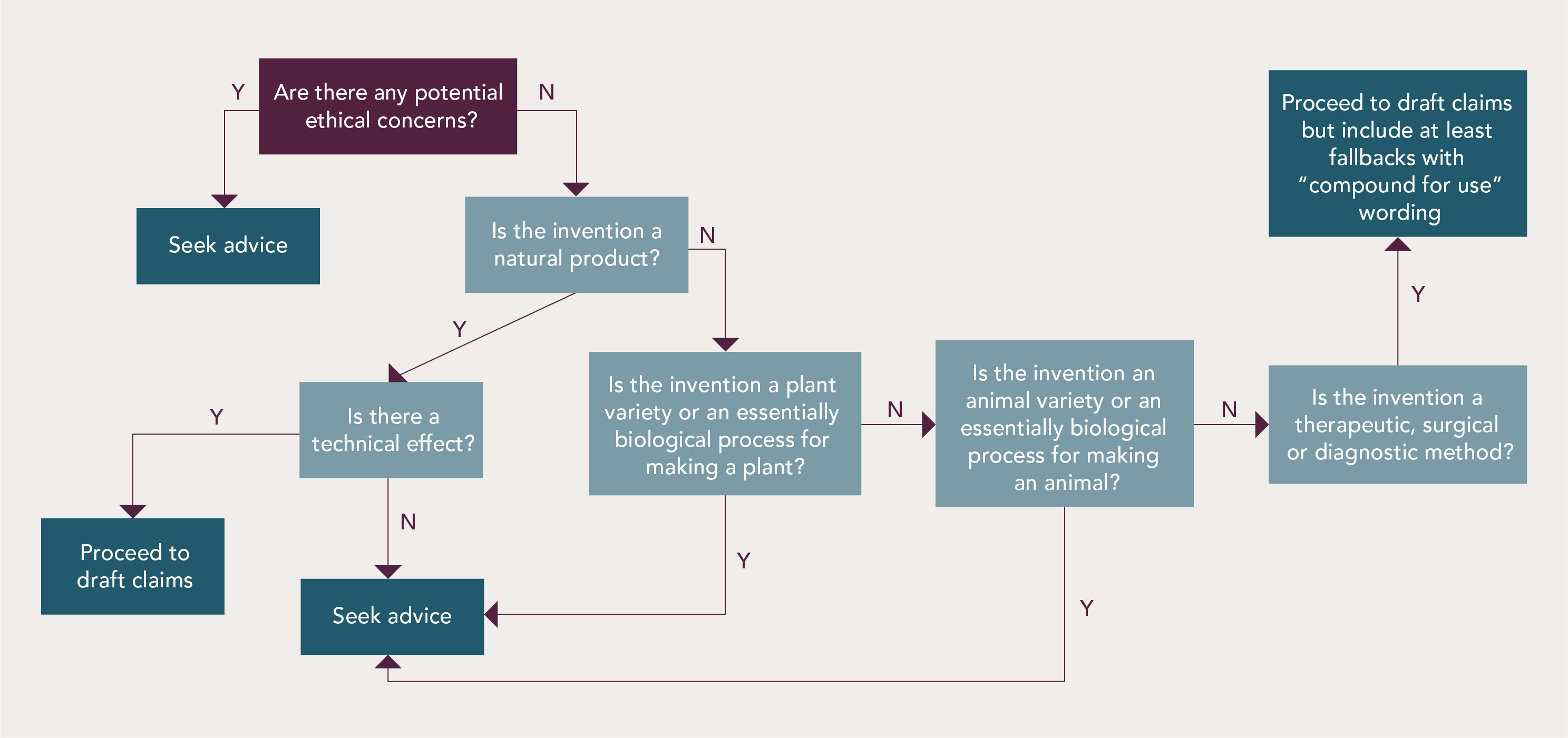
In the life sciences field, most subject-matter eligibility problems can be overcome by incorporating a few minor adjustments at the drafting stage. The flowchart below summarizes the steps applicants can take themselves and when advice from a European attorney is recommended.
For more information on any of the topics in this article, please contact Victoria or Maeve.
[1]Article 53(a) EPC
[2] Rule 28(1) EPC
[3]Article 53(b) EPC; Rule 26(4) EPC; Rule 28(2) EPC; Rule 26(5) EPC
[4]For more detail, please see our earlier blog post here reporting this Decision.
[5]Article 52(2)(a) EPC
[6]see G-II, 3.1
[7]Rule 29 EPC
[8]Article 53(c) EPC
[9]T 1635/09
[10]Contraceptive method claims have also been rejected by the EPO for being private and personal and thus not “an industrial activity”. The case in question, T 74/93, is an interesting read; the applicant’s defense was that prostitution is the oldest profession in the world and that, consequently, contraceptive methods were capable of industrial activity.
Related Offices
Contacts

- Maeve O'Flynn
- Partner
- London
- +44 (0)20 7864 2856
Copyright © 2021 Finnegan, Henderson, Farabow, Garrett & Dunner, LLP.
DISCLAIMER: Although we wish to hear from you, information exchanged in this blog cannot and does not create an attorney-client relationship. Please do not post any information that you consider to be personal or confidential. If you wish for Finnegan, Henderson, Farabow, Garrett & Dunner, LLP to consider representing you, in order to establish an attorney-client relationship you must first enter a written representation agreement with Finnegan. Contact us for additional information. One of our lawyers will be happy to discuss the possibility of representation with you. Additional disclaimer information.


